DKV health insurance for individuals
Health insurance that offers access to the best private medicine service
-
We will take care of your physical and emotional health.
-
We make all the paperwork easy.
Take out a policy now with a 35% discount.
Discounts for 5 years.


Why choose health insurance with DKV?
Extensive experience
More than 2 million customers put their trust in DKV. We have been taking care of your health for over 25 years.
Doctors and centres
Más de 51,000 médicos y 1,000 centros concertados, 23 centros propios y 93 de los 100 mejores hospitales.
24-hour assistance
Medical chat and video consultation with specialists wherever you are, offering you the best assistance.
Online administrative procedures
You can request authorisations and medical appointments, access your medical reports or process reimbursements through the app.
Calculate your medical insurance
Fill in the following form:
Doctors, centres an own health spaces
Más de 51,000 médicos y 1,000 centros de salud a tu disposición.
DKV medical directory
El valor de un seguro médico está en su cuadro médico. Con DKV cuentas con más de 51,000 profesionales y 1,000 centros en España, atención rápida y la tranquilidad de elegir siempre al especialista que prefieras en cada momento.
Medical specialities
-
Complete Medical Directory
See medical directory -
Medical specialists
Consultas desde el primer día con medicina general y todas las especialidades: ginecología, pediatría, traumatología, dermatología, alergología y más.
-
Clinical analyses
Pruebas de laboratorio (sangre, orina, heces) para diagnósticos rápidos y precisos.
-
Diagnostic tests
Radiologías, ecografías y revisiones preventivas prescritas por tu especialista.
-
Dental treatments
Dental cover included: limpiezas, extracciones y servicios con descuento en ortodoncia y estética dental.
-
Health and wellness services
Access to DKV Club Salud y Bienestar con precios especiales por ser cliente de DKV en nutrición, deporte, fertilidad y bienestar.
Espacios de Salud DKV
-
Doctors and centres
Elige entre una amplia red hospitalaria y especialistas de referencia en España, incluidos 93 de los 100 mejores hospitales.
-
Espacios de Salud DKV
Whether you are a DKV customer or not, we have our own Espacios de Salud propios con especialistas y odontología, tecnología avanzada y atención personalizada.
-
Online consultation with specialists
Videoconsultas ilimitadas, chat médico 24h y receta electrónica desde tu móvil.
-
Digital midwife and Health coach
Acompañamiento en embarazo y posparto, y asesoramiento para mejorar alimentación, deporte y hábitos saludables.
-
Medical helpline
24-Hour DKV Physician wherever you are, as well as Specialised medical lines: pediatría, mujer, nutrición y apoyo emocional.
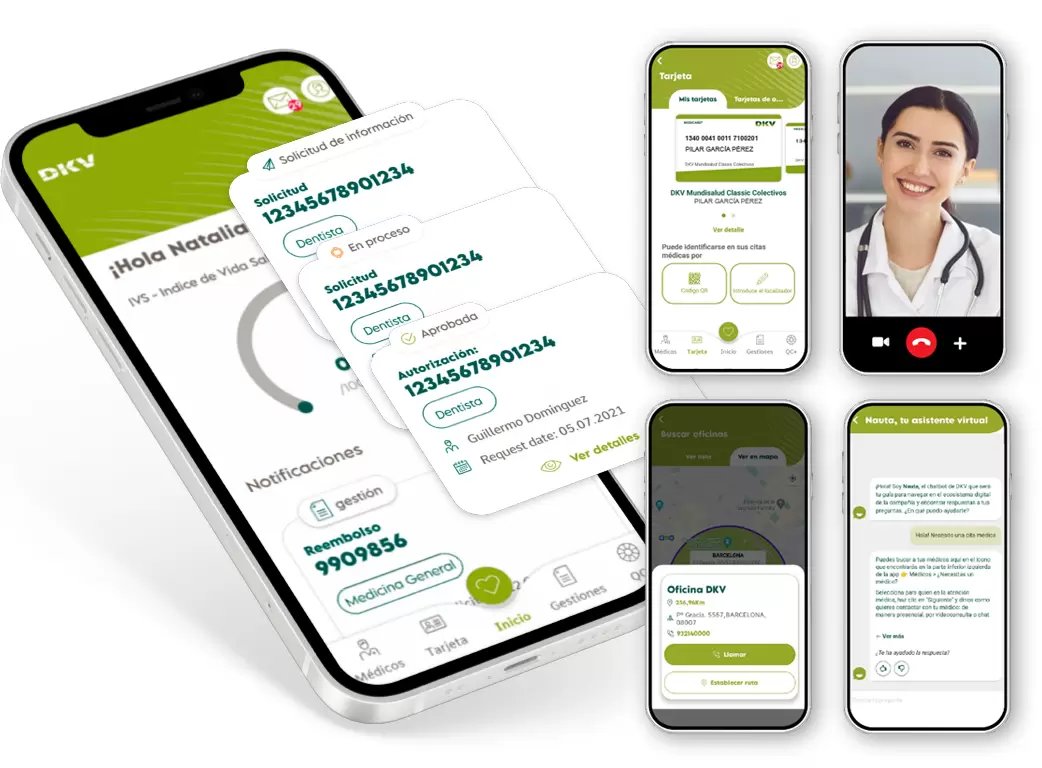
Your insurance always with you
Manage your insurance and access digital health services from anywhere with the Activa DKV app.
Do you need help?
If you are thinking about taking out an insurance policy and you have questions, we will answer them in the way you choose.